What Color Is Benedict's Solution
Benedict's Test is used to test for simple carbohydrates. The Benedict's test identifies reducing sugars (monosaccharide'south and some disaccharides), which have gratis ketone or aldehyde functional groups. Benedict'southward solution can be used to exam for the presence of glucose in urine.
Some sugars such every bit glucose are called reducing sugars because they are capable of transferring hydrogens (electrons) to other compounds, a procedure called reduction. When reducing sugars are mixed with Benedicts reagent and heated, a reduction reaction causes the Benedicts reagent to modify color. The colour varies from green to dark red (brick) or rusty-brownish, depending on the amount of and blazon of carbohydrate.
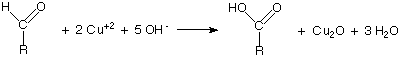
Bridegroom'due south quantitative reagent contains potassium thiocyanate and is used to make up one's mind how much reducing carbohydrate is present. This solution forms a copper thiocyanate precipitate which is white and tin can be used in a titration. The titration should be repeated with 1% glucose solution instead of the sample for calibration
Principle of Benedict'southward Exam
When Benedict's solution and unproblematic carbohydrates are heated, the solution changes to orange reddish/ brick red. This reaction is caused by the reducing property of simple carbohydrates. The copper (II) ions in the Benedict's solution are reduced to Copper (I) ions, which causes the color change.
The cerise copper(I) oxide formed is insoluble in water and is precipitated out of solution. This accounts for the precipitate formed. Equally the concentration of reducing sugar increases, the nearer the final color is to brick-red and the greater the precipitate formed. Sometimes a brick red solid, copper oxide, precipitates out of the solution and collects at the bottom of the exam tube.
Sodium carbonate provides the alkaline weather condition which are required for the redox reaction.Sodium citrate complexes with the copper (II) ions then that they do not deteriorate to copper(I) ions during storage.
Complex carbohydrates such as starches Exercise NOT react positive with the Benedict's test unless they are broken down through heating or digestion (try chewing crackers then doing the exam). Table sugar (disaccharide) is a non-reducing sugar and does also not react with the iodine or with the Bridegroom Reagent. Carbohydrate needs to exist decomposed into its components glucose and fructose then the glucose test would be positive only the starch examination would even so exist negative.
Composition and Training of Benedict'due south Solution
Bridegroom's solution is a deep-blue element of group i solution used to exam for the presence of the aldehyde functional group, – CHO.
Anhydrous sodium carbonate = 100 gm
Sodium citrate – 173 gm
Copper(2) sulfate pentahydrate = 17.3 gm
One litre of Benedict's solution can be prepared from 100 thousand of anhydrous sodium carbonate, 173 g of sodium citrate and 17.3 g of copper(Ii) sulfate pentahydrate.
Process of Benedict's Examination
- Approximately i ml of sample is placed into a clean test tube.
- 2 ml (10 drops) of Bridegroom's reagent (CuSO4) is placed in the test tube.
- The solution is then heated in a boiling water bath for 3-v minutes.
- Observe for color change in the solution of exam tubes or precipitate formation.
Result Estimation of Benedict'south Test
If the color upon boiling is changed into green, then there would exist 0.one to 0.five percent saccharide in solution.
If it changes color to yellow, so 0.5 to 1 percent sugar is present.
If information technology changes to orange, then it means that ane to 1.5 pct sugar is present.
If colour changes to cerise,then 1.5 to 2.0 percent sugar is present.
And if color changes to brick reddish,it ways that more than 2 per centum sugar is present in solution.

Positive Bridegroom'south Exam: Formation of a ruby-red precipitate inside 3 minutes. Reducing sugars present. Case: Glucose
Negative Bridegroom's Test: No colour change (Remains Blue). Reducing sugars absent. Example: Sucrose.
References
- National Institutes of Health, Testing for Lipids, Proteins and Carbohydrates- Benedict's solution.
- Fayetteville State University- Biological Molecules: Carbohydrates, Lipids, Proteins.
- Harper Higher- Benedict'southward Test.
- National Biochemicals Corp.- Bridegroom'S SOLUTION (MB4755).
- Scientific discipline Olympiad- Use of Benedict'due south Solution.
- Vivid Biological science Pupil 2015- Food Tests- Benedict'due south Test for Reducing Sugars.
- BBC Bitesize- Chemistry- Carbohydrates.
- University of Manitoba- The Molecules of Life: Biochemistry- Carbohydrates.
- Northern Kentucky Academy- Bridegroom'south Reagent: A Examination for Reducing Sugars.
- KNUST Open Educational Resource, Benedict's Test – Qualitative Test in Carbohydrates.
- Mark Rothery's Biology Web Site- Biochemical Tests.
- All Medical Stuff- Benedict's test for reducing sugar.
- Hendrix College- Benedicts Exam for Glucose.
- Info Please- Bridegroom'southward solution.
- Mystrica- Benedict's Test.
- Amrita Virtual Lab Collaborative Platform- Qualitative Analysis of Carbohydrates.
- Wikipedia.
Similar Posts:
- Oxidase Test- Principle, Uses, Procedure, Types, Outcome Estimation, Examples and Limitations
- Indole Examination- Principle, Reagents, Procedure, Outcome Interpretation and Limitations
- The Triple Carbohydrate Iron (TSI) Test – Principle, Procedure, Uses and Interpretation
- Bile Solubility Test- Principle, Reagents, Procedure and Event Interpretation
What Color Is Benedict's Solution,
Source: https://microbiologyinfo.com/benedicts-test-principle-composition-preparation-procedure-and-result-interpretation/
Posted by: brambletiont1992.blogspot.com


0 Response to "What Color Is Benedict's Solution"
Post a Comment